:max_bytes(150000):strip_icc()/phase-changes-56a12ddd3df78cf772682e07.png)
For example, the melting of ice to give water is a physical change because water only changes its physical state from solid to liquid without affecting its chemical identity.Ī chemical change is a process in which the substance changes its chemical identity or composition. Properties of matter Are the properties observed/measured without changing the chemical identity of the substance?įlammability - when wood burns it changes to ashesĬorrosion - rusting of iron objects after exposure to moist air for a long timeĭecomposition - hydrogen peroxide decomposes into water and oxygen in the presence of heat or lightĬhanges in the matter are classified into two categories: physical change and chemical change.Ī physical change is when the substance changes its physical appearance or state without affecting the chemical identity. Properties of matter can be classified into two types depending on whether the particular property is determined by changing the chemical identity or composition of the substance. The change of gas to liquid is known as condensation.The change of liquid to solid is known as freezing.The change of solid to gas is known as sublimation.The change of gas to solid is known as deposition.The change of liquid to gas is known as vaporization.The change of solid to liquid is known as melting. 
State changes have different names depending on which states are involved: Water is one of the few substances which can exist in all three states: solid ice, liquid water, gaseous steam. A free teacher account also allows you to create playlists of games and assignments for students and track class progress.The conversion of a substance from one state to another is known as the change of state. You can access all of the games on Legends of Learning for free, forever, with a teacher account. 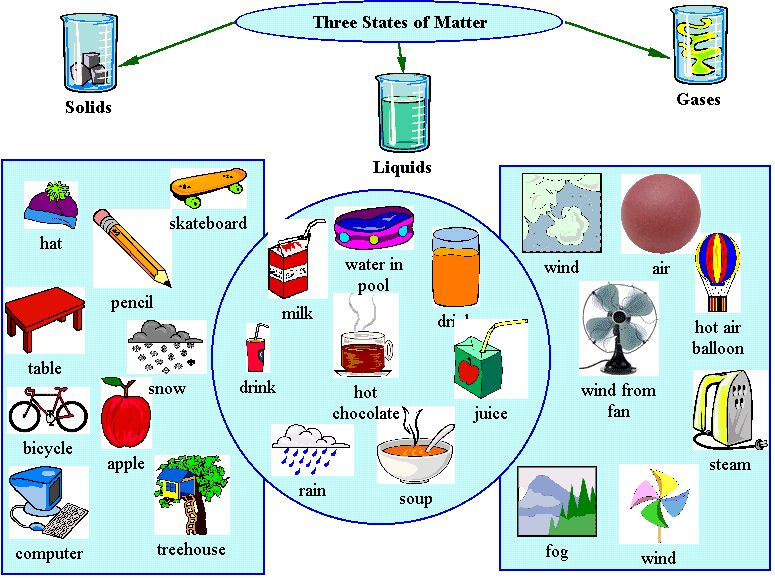
For some types of rock, decreasing pressure can also cause them to melt.Ī preview of each game in the learning objective is found below. Decreasing pressure can cause it to vaporize. When the pressure exerted on a substance increases, it can cause the substance to condense. When energy is removed, the opposite happens, decreasing the substance’s temperature and turning it from liquid to solid (freezing), gas to solid (deposition), or from gas to liquid (condensation). When thermal energy is added to a substance, its temperature increases, which can change its state from solid to liquid (melting), liquid to gas (vaporization), or solid to gas (sublimation). Both temperature and pressure can be measured, and state changes can be observed. Physical conditions like temperature and pressure affect state of matter. Gases expand or contract to fill the available space, meaning they don’t maintain their shape or volume.Ī substance’s state of matter is an extrinsic property, meaning it can be changed by its environment. They move quickly relative to each other. Gas – particles do not stay in contact, although they can collide.Liquids change shape based on their container, but maintain their volume. Liquid – particles stay in contact, but they are able to move relative to each other.Solid – particles stay in contact with each other, vibrating in place but not moving relative to each other, maintaining the solid’s shape and volume.Particles are in constant motion, but they interact differently depending on the state of matter. A substance’s state of matter - solid, liquid, gas, or plasma - depends on how its molecules move and maintain their volume and shape. Concepts CoveredĪtoms and molecules are the particles that make up matter. Scroll down for a preview of this learning objective’s games and the concepts they drive home. The Effects of Temperature and Pressure on State learning objective - based on NGSS and state standards - delivers improved student engagement and academic performance in your classroom, as demonstrated by research. In this series of games, your students will learn how and why substances undergo phase changes.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
